- Slide show, p1
- References, p2
References:
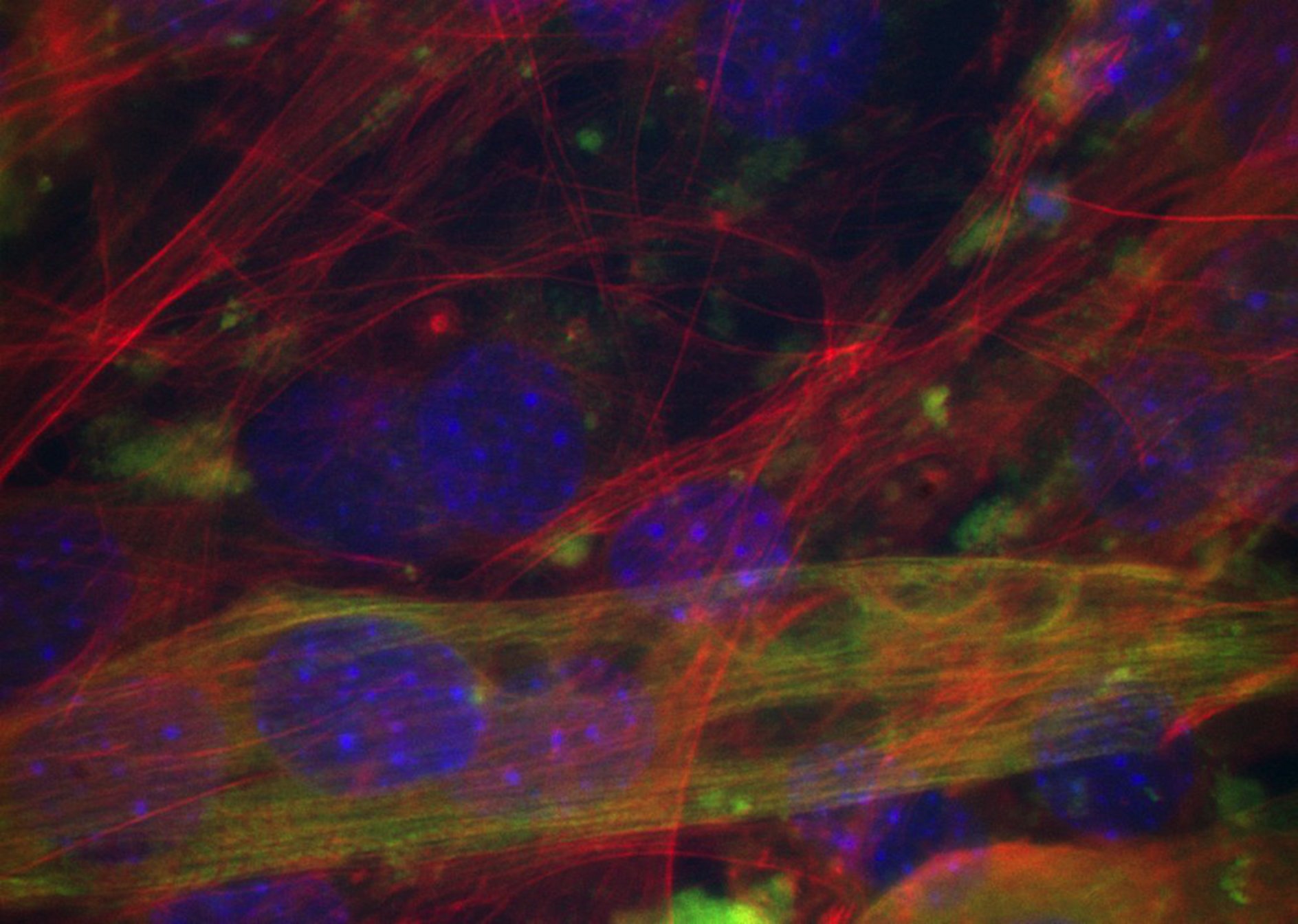
![]() NiceCells (Fluorescent Cell - Tubulin labeling (green)) Image source : slide through the courtesy of Angelica Keller, image acquisition from the author. BH2 Olympus microscope and Scion CCD CFW-1310M Camera. Project link.
NiceCells (Fluorescent Cell - Tubulin labeling (green)) Image source : slide through the courtesy of Angelica Keller, image acquisition from the author. BH2 Olympus microscope and Scion CCD CFW-1310M Camera. Project link.
![]() Neuron - before & after deconvolution: mouse hyppocampal neuron culture in conditioned medium. Two channels fluorescence laser scanning confocal image stack acquisition. Red; postsynaptic elements PSD95 staining, green; presynaptic elements vGlut staining. Left, maximum projection of the initial signal. Right, maximum projection after deconvolution of the z stack. Olympus IX81 microscope, FV1000. Culture and immuno-staining author: Olivier Stettler. Image acquisition and processing: Gilles Carpentier. Contact for biological aspects: Pr. Olivier Stettler. Related project.
Neuron - before & after deconvolution: mouse hyppocampal neuron culture in conditioned medium. Two channels fluorescence laser scanning confocal image stack acquisition. Red; postsynaptic elements PSD95 staining, green; presynaptic elements vGlut staining. Left, maximum projection of the initial signal. Right, maximum projection after deconvolution of the z stack. Olympus IX81 microscope, FV1000. Culture and immuno-staining author: Olivier Stettler. Image acquisition and processing: Gilles Carpentier. Contact for biological aspects: Pr. Olivier Stettler. Related project.
![]() Nice-neuron-synapses: mouse hyppocampal neuron culture in conditioned medium. Three channels fluorescence laser scanning confocal image acquisition. Blue; DNA, red ; postsynaptic elements PSD95 staining, green; presynaptic elements vGlut staining. Olympus IX81 microscope, FV1000. Culture and immuno-staining Olivier Stettler. Image acquisition and processing: Gilles Carpentier. Contact for biological aspects: Pr. Olivier Stettler. Related project.
Nice-neuron-synapses: mouse hyppocampal neuron culture in conditioned medium. Three channels fluorescence laser scanning confocal image acquisition. Blue; DNA, red ; postsynaptic elements PSD95 staining, green; presynaptic elements vGlut staining. Olympus IX81 microscope, FV1000. Culture and immuno-staining Olivier Stettler. Image acquisition and processing: Gilles Carpentier. Contact for biological aspects: Pr. Olivier Stettler. Related project.
![]() Neuron synaptic area detection - user selection: mouse hyppocampal neuron culture in conditioned medium. Two channels fluorescence laser scanning confocal image stack acquisition. Red ; postsynaptic elements PSD95 staining, green; presynaptic elements vGlut staining, user selection in blue. Maximum projection after deconvolution of a z stack. Olympus IX81 microscope, FV1000. Culture and immuno-staining author: Olivier Stettler. Image acquisition and processing: Gilles Carpentier. Contact for biological aspects: Pr. Olivier Stettler. Related project.
Neuron synaptic area detection - user selection: mouse hyppocampal neuron culture in conditioned medium. Two channels fluorescence laser scanning confocal image stack acquisition. Red ; postsynaptic elements PSD95 staining, green; presynaptic elements vGlut staining, user selection in blue. Maximum projection after deconvolution of a z stack. Olympus IX81 microscope, FV1000. Culture and immuno-staining author: Olivier Stettler. Image acquisition and processing: Gilles Carpentier. Contact for biological aspects: Pr. Olivier Stettler. Related project.
![]() Neuron synaptic area detection: mouse hyppocampal neuron culture in conditioned medium. Two channels fluorescence laser scanning confocal image stack acquisition. Red ; postsynaptic elements PSD95 staining, green presynaptic elements vGlut staining. Maximum projection after deconvolution of a z stack following by automatic detection of synaptic areas (yellow) in user selection (previous image), by using an original program. Olympus IX81 microscope, FV1000. Culture and immuno-staining author: Olivier Stettler. Image acquisition, processing and analysis: Gilles Carpentier. Contact for biological aspects: Pr. Olivier Stettler. Related project.
Neuron synaptic area detection: mouse hyppocampal neuron culture in conditioned medium. Two channels fluorescence laser scanning confocal image stack acquisition. Red ; postsynaptic elements PSD95 staining, green presynaptic elements vGlut staining. Maximum projection after deconvolution of a z stack following by automatic detection of synaptic areas (yellow) in user selection (previous image), by using an original program. Olympus IX81 microscope, FV1000. Culture and immuno-staining author: Olivier Stettler. Image acquisition, processing and analysis: Gilles Carpentier. Contact for biological aspects: Pr. Olivier Stettler. Related project.
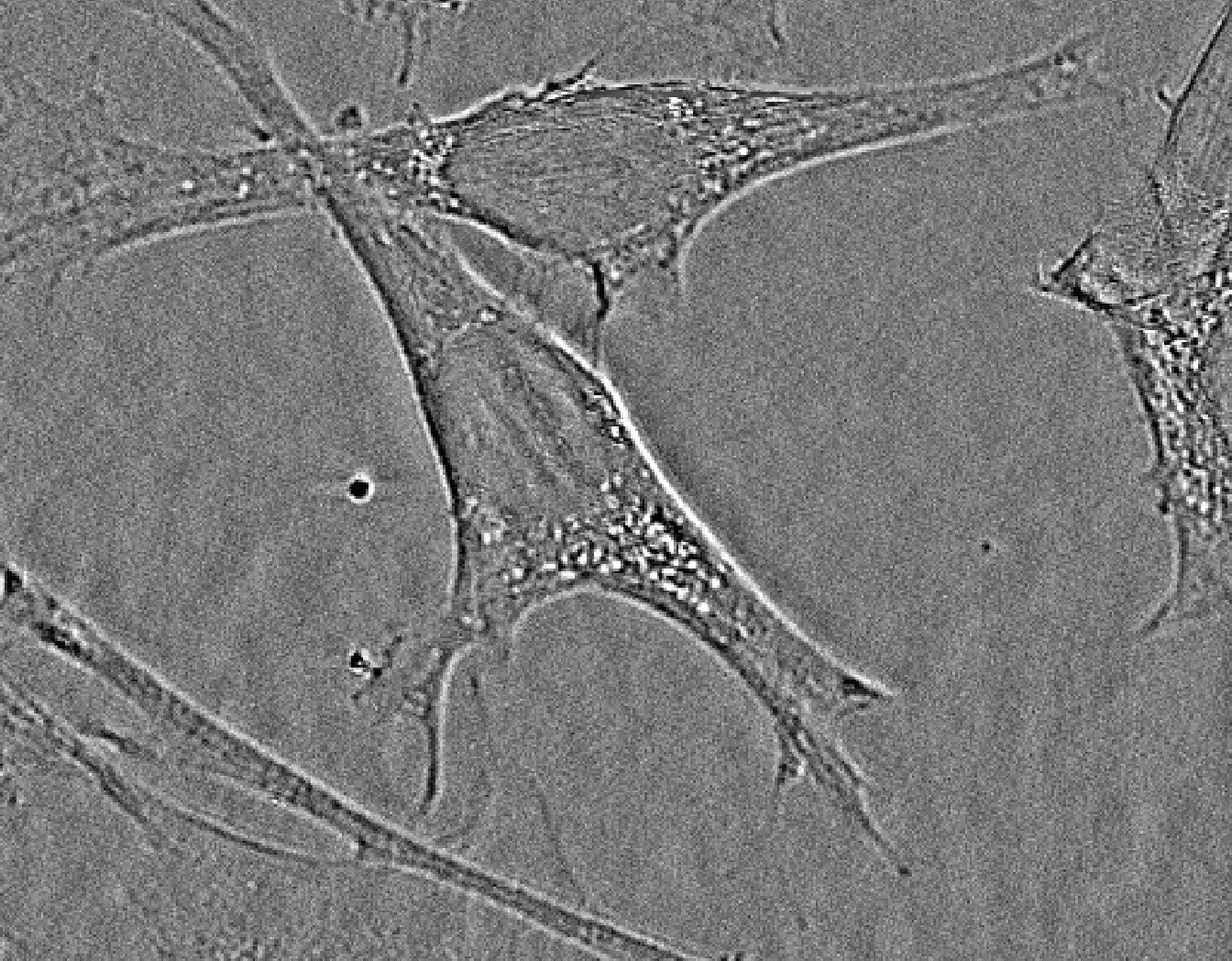
![]() Human macrophage derived monocytes-PH and Human macrophage derived monocytes-PH-ROS-DNA: human macrophage derived monocyte culture. Respectively, phase contrast acquisition and phase contrast plus two channels fluorescence acquisition (ROS, red and DNA, blue) Olympus IX81 Microscope. Culture author and image acquisition: Nicolas Rebergue. Contact for biological aspects: Gael Le Douaron / Alexandre Fifre.
Human macrophage derived monocytes-PH and Human macrophage derived monocytes-PH-ROS-DNA: human macrophage derived monocyte culture. Respectively, phase contrast acquisition and phase contrast plus two channels fluorescence acquisition (ROS, red and DNA, blue) Olympus IX81 Microscope. Culture author and image acquisition: Nicolas Rebergue. Contact for biological aspects: Gael Le Douaron / Alexandre Fifre.
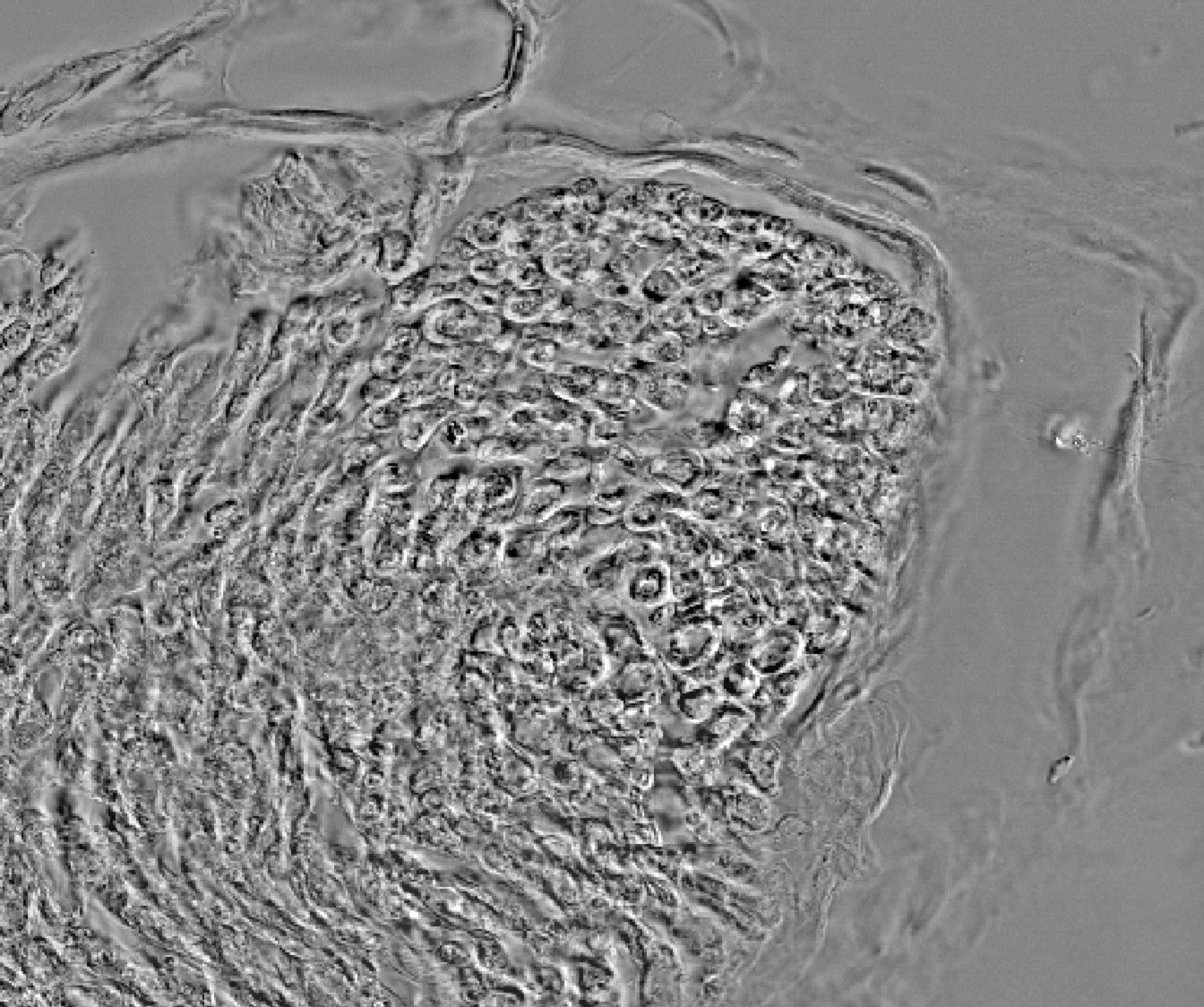
![]() Human macrophage derived monocytes-PH-ROS-DNA-An: human macrophage derived monocyte culture. Phase contrast and two channels fluorescence acquisition (ROS, red and DNA, blue) following by automatic detection and analysis, Olympus IX80 Microscope. Culture author and image acquisition: Nicolas Rebergue. Contact: Gael Le Douaron / Alexandre Fifre. Image processing and analysis: Gilles Carpentier.
Human macrophage derived monocytes-PH-ROS-DNA-An: human macrophage derived monocyte culture. Phase contrast and two channels fluorescence acquisition (ROS, red and DNA, blue) following by automatic detection and analysis, Olympus IX80 Microscope. Culture author and image acquisition: Nicolas Rebergue. Contact: Gael Le Douaron / Alexandre Fifre. Image processing and analysis: Gilles Carpentier.
![]() HUVEC lens free microscopy - raw. Lensfree raw acquisition of human umbilical vein endothelial cells cultured on Matrigel. The field of view is 29.4 mm2. Details in below references. Related project.
HUVEC lens free microscopy - raw. Lensfree raw acquisition of human umbilical vein endothelial cells cultured on Matrigel. The field of view is 29.4 mm2. Details in below references. Related project.
![]() HUVEC lens free microscopy - reconstructed. Holographic reconstruction of the recorded image shown in (a). The results is a modulus image which depicts the cells in dark gray over a light grey background.
HUVEC lens free microscopy - reconstructed. Holographic reconstruction of the recorded image shown in (a). The results is a modulus image which depicts the cells in dark gray over a light grey background.
The holographic reconstruction consists first in back-propagating the light intensity recorded by the sensor through the Fresnel function. The recovered complex image is however strongly affected by the so-called ‘twin image’, an artefact that results from a lack of phase information in the acquisition process. To retrieve the phase in the sensor plane and consequently diminish the ‘twin-image’ artefact in the reconstructed images, we used a phase-retrieval algorithm. The purpose of such an algorithm is to recover the complex image of the sample from the phase-less recorded holographic image. It allows to refine the estimate of the complex image in the sensor plane by iteratively applying update rules in a gradient descent scheme.
Credits : Cell culture: F.Navarro (ab), M. Menneteau (ab), Holographic reconstruction: L. Hervé (ab), C. Allier (ab), a Univ. Grenoble Alpes, F‐38000 Grenoble, France. b CEA LETI MlNATEC Campus, F-38054 Grenoble, France.
![]() HUVEC lens free microscopy-analyzed: Lens free microscopy Image analyzed by a customized version of the Angiogenesis Analyzer after holographic reconstruction. Details in above references.
HUVEC lens free microscopy-analyzed: Lens free microscopy Image analyzed by a customized version of the Angiogenesis Analyzer after holographic reconstruction. Details in above references.
![]() Microcosme: Fluorescent algae and other microorganisms. Olympus IX80 microscope coupled to an ORCA R2 Hamamatsu CCD camera. Sample source; Iglika Christova, image acquisition and processing, Gilles Carpentier. Project link.
Microcosme: Fluorescent algae and other microorganisms. Olympus IX80 microscope coupled to an ORCA R2 Hamamatsu CCD camera. Sample source; Iglika Christova, image acquisition and processing, Gilles Carpentier. Project link.
![]() HUVEC Lensfree Microscopy, and HUVEC Lensfree Microscopy + Analysis. HUVEC network acquired by lens free microscopy and analyzed by a cutomized version of the Angiogenesis Analyzer. Details and timelapse here.
HUVEC Lensfree Microscopy, and HUVEC Lensfree Microscopy + Analysis. HUVEC network acquired by lens free microscopy and analyzed by a cutomized version of the Angiogenesis Analyzer. Details and timelapse here.
![]() HUVEC Phase Contrast, HUVEC Phase Contrast + Analysis, HUVEC Fluo, HUVEC Fluo + Analysis, are HUVEC networks analyzed with the Angiogenesis Analyzer.
HUVEC Phase Contrast, HUVEC Phase Contrast + Analysis, HUVEC Fluo, HUVEC Fluo + Analysis, are HUVEC networks analyzed with the Angiogenesis Analyzer.
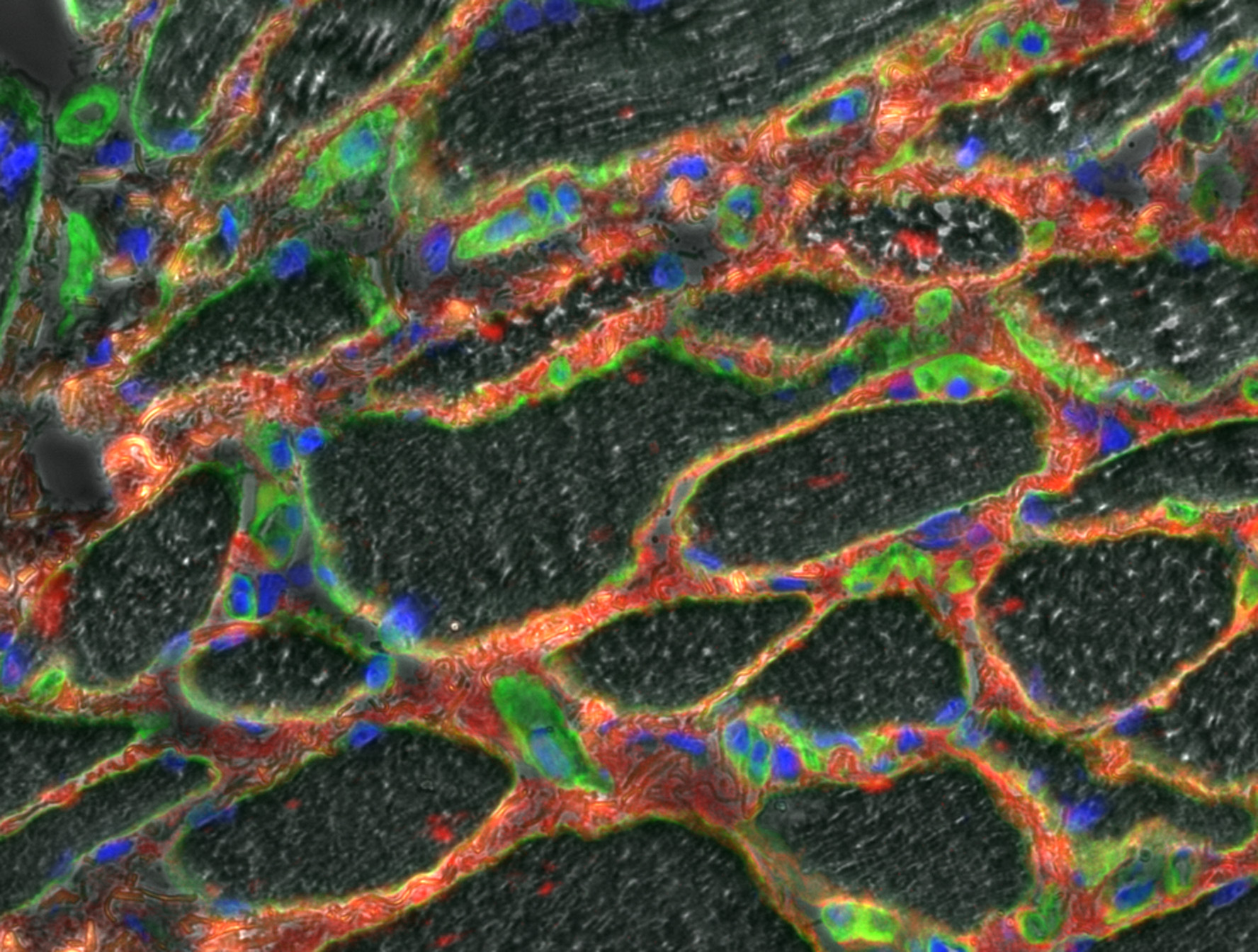
![]() HistoMuscle-4Channels and HistoMuscle-3Channels : Muscle slice; 3 channels immunolabeling from Elisa Negroni, image acquisition from Emilie Henault. IX81 inverted Olympus microscope and Orca Hamamatsu R2 CCD camera. Project link.
HistoMuscle-4Channels and HistoMuscle-3Channels : Muscle slice; 3 channels immunolabeling from Elisa Negroni, image acquisition from Emilie Henault. IX81 inverted Olympus microscope and Orca Hamamatsu R2 CCD camera. Project link.
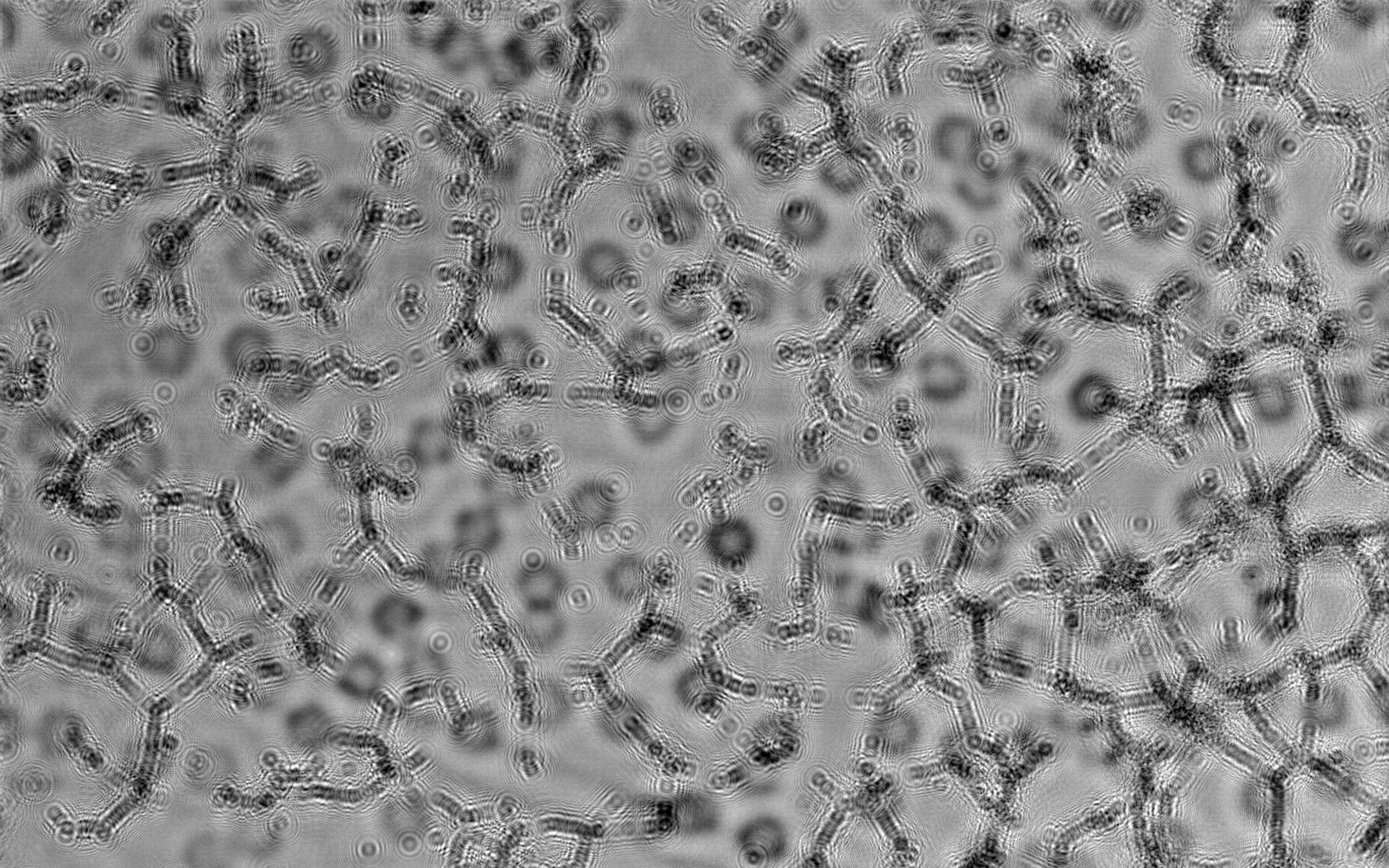
![]() Myoblast - LSM transmission, Myoblast - LSM transmission-Deblured and Myoblast - LSM transmission-Deblured-fft : different improvements of a laser scanning transmission image by using composite unsharp mask and fast Fourier transform. Transmission laser scanning microscopy image from the author. Project link.
Myoblast - LSM transmission, Myoblast - LSM transmission-Deblured and Myoblast - LSM transmission-Deblured-fft : different improvements of a laser scanning transmission image by using composite unsharp mask and fast Fourier transform. Transmission laser scanning microscopy image from the author. Project link.
![]() Rat sciatic nerve and Rat sciatic nerve-deblurred : Images before and after deblurring. Initial image from the courtesy of Dr Brigitte Blondet. Device for the image acquisition: monochrome CMOS Sound Vision camera mounted onto an Olympus BH-2 microscope. Project link.
Rat sciatic nerve and Rat sciatic nerve-deblurred : Images before and after deblurring. Initial image from the courtesy of Dr Brigitte Blondet. Device for the image acquisition: monochrome CMOS Sound Vision camera mounted onto an Olympus BH-2 microscope. Project link.
![]() 3 channel fluorescence. The example contains a triple labeling of a differentiated mouse myogenic cell line. Cell culture and immunochemistry; Juliette Peltzer, images from the courtesy of Dr Angelica Keller. Project links, here and here.
3 channel fluorescence. The example contains a triple labeling of a differentiated mouse myogenic cell line. Cell culture and immunochemistry; Juliette Peltzer, images from the courtesy of Dr Angelica Keller. Project links, here and here.
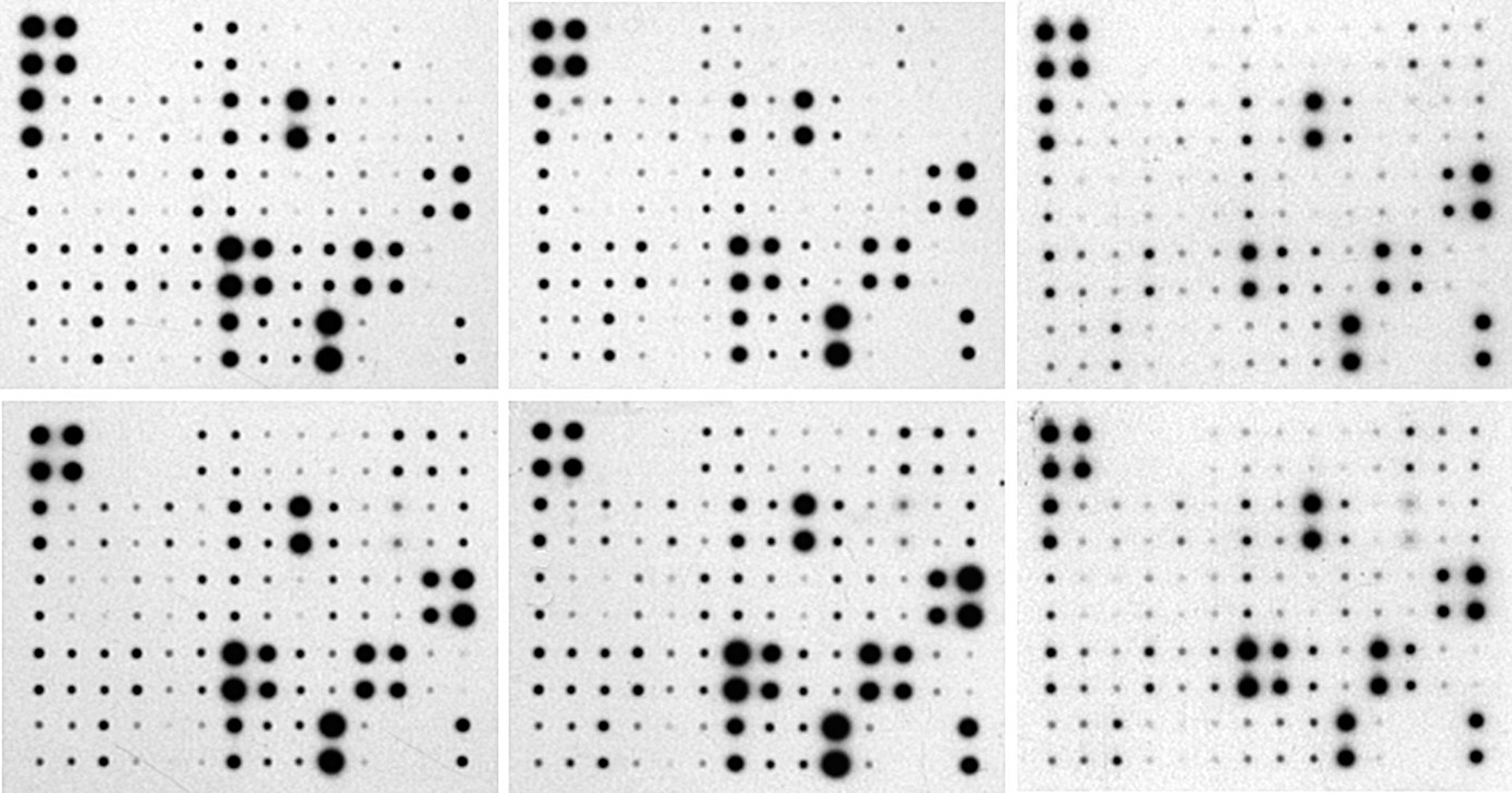
![]() « Protein array » and « Analyzed protein array » are protein arrays analyzed by the Protein Array Analyzer, ImageJ conference.
« Protein array » and « Analyzed protein array » are protein arrays analyzed by the Protein Array Analyzer, ImageJ conference.
![]() Whole mouse retina. Whole retina vasculature. Images obtained through an Orca R2 Hamamatsu CCD camera mounted on an IX 81 Olympus microscope (Obj 5x). About 9 images assembled to build full retina pictures, by using the MosaicJ plugin. Experiment and image acquisition author: Dr. Melissande Cossuta; image analysis : Gilles Carpentier. News.
Whole mouse retina. Whole retina vasculature. Images obtained through an Orca R2 Hamamatsu CCD camera mounted on an IX 81 Olympus microscope (Obj 5x). About 9 images assembled to build full retina pictures, by using the MosaicJ plugin. Experiment and image acquisition author: Dr. Melissande Cossuta; image analysis : Gilles Carpentier. News.
![]() Whole mouse retina - analysis. Whole mouse retina after vasculature analysis in user selections (customized Angiogenesis Analyzer). Experiment and image acquisition author: Dr. Melissande Cossuta; image analysis : Gilles Carpentier. News.
Whole mouse retina - analysis. Whole mouse retina after vasculature analysis in user selections (customized Angiogenesis Analyzer). Experiment and image acquisition author: Dr. Melissande Cossuta; image analysis : Gilles Carpentier. News.
![]() Whole mouse retina - modeled vasculature. Modeled map of mouse retina vasculature; meshes (cyan), segments (magenta), branches (green) and junctions (blue & red). Obtained from a customized Angiogenesis Analyzer. Experiment and image acquisition author: Dr. Melissande Cossuta; image analysis : Gilles Carpentier. News.
Whole mouse retina - modeled vasculature. Modeled map of mouse retina vasculature; meshes (cyan), segments (magenta), branches (green) and junctions (blue & red). Obtained from a customized Angiogenesis Analyzer. Experiment and image acquisition author: Dr. Melissande Cossuta; image analysis : Gilles Carpentier. News.
![]() Mouse retina sample. Mouse retina vasculature sample before analysis by customized Angiogenesis Analyzer. Experiment and image acquisition author: Experiment and image acquisition author: Dr. Melissande Cossuta; image analysis : Gilles Carpentier. News.
Mouse retina sample. Mouse retina vasculature sample before analysis by customized Angiogenesis Analyzer. Experiment and image acquisition author: Experiment and image acquisition author: Dr. Melissande Cossuta; image analysis : Gilles Carpentier. News.
![]() Mouse retina analyzed sample. Mouse retina vasculature sample after analysis by customized Angiogenesis Analyzer. Experiment and image acquisition author: Dr. Melissande Cossuta; image analysis : Gilles Carpentier. News.
Mouse retina analyzed sample. Mouse retina vasculature sample after analysis by customized Angiogenesis Analyzer. Experiment and image acquisition author: Dr. Melissande Cossuta; image analysis : Gilles Carpentier. News.
![]() Fibrin Bead Assay : Simple environment. Initial image. Experiment and image acquisition author: Dr. Sarah Berndt; image analysis : Gilles Carpentier. Related links: Angiogenesis Analyzer, ImageJ User and Developer Conference 2012 poster and short article of proceedings, Scientific Report article.
Fibrin Bead Assay : Simple environment. Initial image. Experiment and image acquisition author: Dr. Sarah Berndt; image analysis : Gilles Carpentier. Related links: Angiogenesis Analyzer, ImageJ User and Developer Conference 2012 poster and short article of proceedings, Scientific Report article.
![]() Fibrin Bead Assay : Simple environment. Microsphere edge detection (red).
Fibrin Bead Assay : Simple environment. Microsphere edge detection (red).
![]() Fibrin Bead Assay : Simple environment. Pseudo microvascular tree detection; segments (magenta), branches (green), nodes (red dots) and junctions (blue).
Fibrin Bead Assay : Simple environment. Pseudo microvascular tree detection; segments (magenta), branches (green), nodes (red dots) and junctions (blue).
![]() Fibrin Bead Assay : Simple environment. Anchorage detection (mauve) and detection of pseudo microvascular tree ; microsphere (red), segments (magenta), branches (green), nodes (red dots) and junctions (blue).
Fibrin Bead Assay : Simple environment. Anchorage detection (mauve) and detection of pseudo microvascular tree ; microsphere (red), segments (magenta), branches (green), nodes (red dots) and junctions (blue).
![]() Fibrin Bead Assay : Complex environment - initial image.
Fibrin Bead Assay : Complex environment - initial image.
![]() Fibrin Bead Assay : Complex environment - Detection of microsphere (red) and pseudo vascular tree; segments (magenta), branches (green), nodes (red dots), junctions (blue) and anchorages (mauve).
Fibrin Bead Assay : Complex environment - Detection of microsphere (red) and pseudo vascular tree; segments (magenta), branches (green), nodes (red dots), junctions (blue) and anchorages (mauve).
![]() Fibrin Bead Assay : Complex environment - Maps of quantified vectorial objects. Microsphere (red) and pseudo vascular tree; segments (Magenta), branches (green), nodes (red dots), junctions (blue) and anchorage (mauve).
Fibrin Bead Assay : Complex environment - Maps of quantified vectorial objects. Microsphere (red) and pseudo vascular tree; segments (Magenta), branches (green), nodes (red dots), junctions (blue) and anchorage (mauve).
![]() Raw RTOVI image. Example of microvascular RTOVI image analysis. Image acquisition author: Dr. Pedro Bastos; image analysis: Gilles Carpentier.
Raw RTOVI image. Example of microvascular RTOVI image analysis. Image acquisition author: Dr. Pedro Bastos; image analysis: Gilles Carpentier.
![]() Segmented RTOVI image. Segmented image coming from a RTOVI recorded movie.
Segmented RTOVI image. Segmented image coming from a RTOVI recorded movie.
![]() Analyzed microvascularity. Analyzed microvascularity organisation of an image coming from a RTOVI recorded movie.
Analyzed microvascularity. Analyzed microvascularity organisation of an image coming from a RTOVI recorded movie.